|
11/22/2023 0 Comments Benzene sigma and pi bonds
That is to say, it is positioned at right angles to those orbitals, with one lobe coming out of the plane of the page and the other going behind the page. 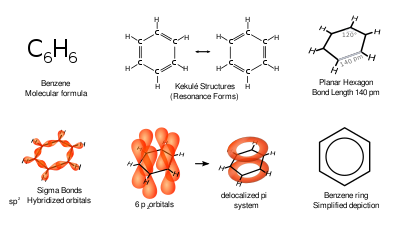
In this top view, the unhybridized p orbital cannot be seen because it also arranges itself to be as far apart from the sp 2 orbitals as possible. A top view of this arrangement is shown below. The ideal angle between sp 2 orbitals is therefore 120 o. That is to say, the carbon nucleus will be at the center of an equilateral triangle, and the three sp 2 orbitals will point to the corners of that triangle. Therefore, the three equivalent sp 2 orbitals will arrange themselves in a trigonal planar configuration. Again, according to VSEPR theory, equivalent orbitals will arrange themselves in 3-D space to be as far apart from each other as possible. It still retains its original energy and shape. The process is shown below.Īs shown, the three resulting sp 2 orbitals are equivalent in energy, but the remaining p orbital has not been affected. We will now reproduce the sp 3 hybridization process for carbon, but instead of taking one s and three p orbitals to make four equivalent sp 3 orbitals, this time we’ll take only one s and two p orbitals to make three equivalent sp 2 orbitals, leaving one p orbital untouched. The second, 's igma star' orbital is higher in energy than the two atomic 1 s orbitals, and is referred to as an antibonding molecular orbital.\) When two atomic 1 s orbitals combine in the formation of H 2, the result is two sigma ( σ) orbitals.Īccording to MO theory, one sigma orbital is lower in energy than either of the two isolated atomic 1 s orbitals –this lower sigma orbital is referred to as a bonding molecular orbital. Mathematical principles tell us that when orbitals combine, the number of orbitals before the combination takes place must equal the number of new orbitals that result from the combination – orbitals don’t just disappear! We saw this previously when we discussed hybrid orbitals: one s and three p orbitals make four sp 3 hybrids. A molecular orbital describes a region of space around two or more atoms inside which electrons are likely to be found. Recall that an atomic orbital (such as the 1s orbital of a hydrogen atom) describes a region of space around a single atom inside which electrons are likely to be found. 
In molecular orbital theory, we make a further statement: we say that the two atomic 1 s orbitals mathematically combine to form two new orbitals. When we described the hydrogen molecule using valence bond theory, we said that the two 1 s orbitals from each atom overlap, allowing the two electrons to be shared and thus forming a covalent bond. Let’s go back and consider again the simplest possible covalent bond: the one in molecular hydrogen (H 2). In order to understand these properties, we will use the ideas of MO theory. Valence bond theory does a remarkably good job at explaining the bonding geometry of many of the functional groups in organic compounds, however, it fails to adequately account for the stability contained in alternating double and single bonds. To understand the source of this stabilization we will use molecular orbital (MO) theory. When we look at carbon-carbon double bonds (C=C), we need to look and see if they are isolated or conjugated. Alternating single and double bonds create a conjugated pi bond system across multiple atoms that lowers the energy and stabilizes the molecule or ion. It is important to train our eye to recognize structural features that have stabilizing effects.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
